Research
Keywords: Pluripotent stem cell, Chimera, Germline
In vitro gametogenesis in various mammals
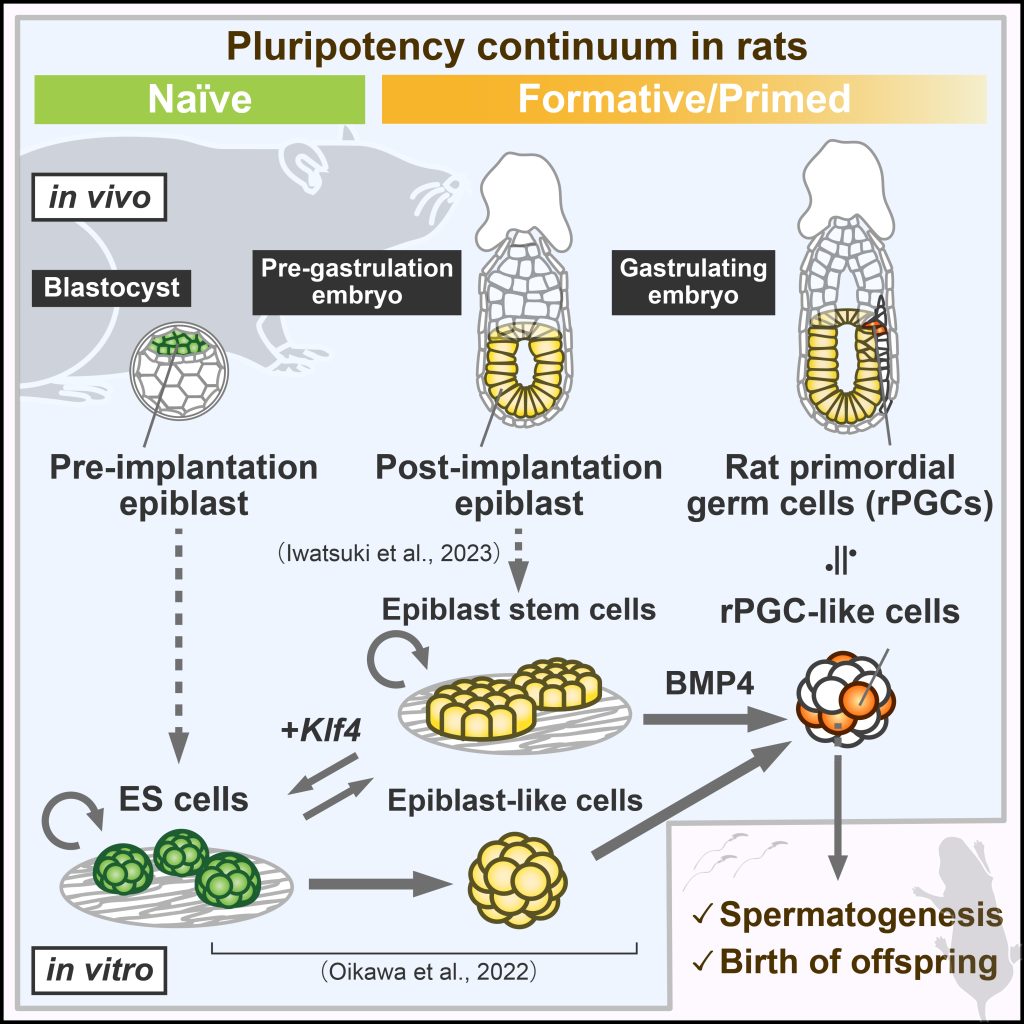
The generation of germ cells from pluripotent stem cells (PSCs) can crucially impact future reproductive medicine and animal breeding. A decade ago, in vitro gametogenesis was established in the mouse. However, the induction of primordial germ cell-like cells (PGCLCs) to produce fertile gametes had not been achieved in any other species. In addition, we demonstrated some key differences between mouse and human germline specification (Kobayashi et al., 2017). In our lab, we aim to develop in vitro systems to induce PGCLCs in various mammals including rats and rabbits (Oikawa et al., 2022, Iwatsuki et al., 2023, Kobayashi et al., 2021, Fig.1-3). Furthermore, by using these systems, we study the conserved or species-specific mechanisms underlying pluripotency transition, germline specification, and subsequent development.
References
Kobayashi T et al., Principles of early human development and germ cell program from conserved model systems. Nature. 2017 Jun 15;546(7658):416-420. doi: 10.1038/nature22812.
Oikawa M et al., Functional primordial germ cell-like cells from pluripotent stem cells in rats. Science. 2022 Apr 8;376(6589):176-179. doi: 10.1126/science.abl4412.
Iwatsuki K et al., Rat post-implantation epiblast-derived pluripotent stem cells produce functional germ cells. Cell Rep Methods. 2023 Jul 27;3(8):100542. doi: 10.1016/j.crmeth.2023.100542.
Kobayashi T et al., Tracing the emergence of primordial germ cells from bilaminar disc rabbit embryos and pluripotent stem cells. Cell Rep. 2021a Oct 12;37(2):109812. doi: 10.1016/j.celrep.2021.109812.
Generation of functional organs via blastocyst complementation
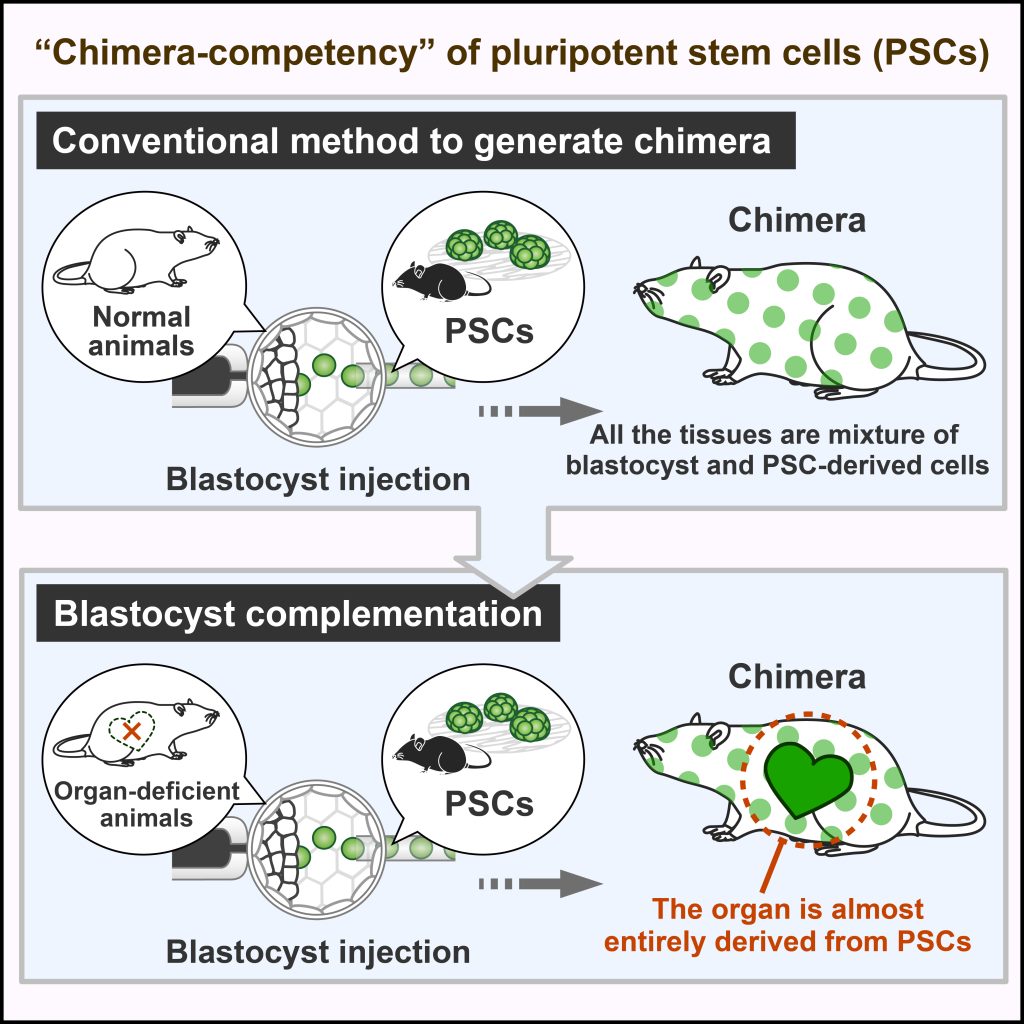
The generation of human organs in model animals allows us not only to provide a disease model that reflects human disease and pathology but also to become a source for organ transplantation in future regenerative medicine. As an approach to achieve it, we have developed the “blastocyst complementation” method which can create an organ in an animal by injecting PSCs into preimplantation embryos derived from organ-deficient animals (Fig. 4 and 5). To demonstrate the proof of principle, we previously demonstrated the successful generation of rat iPSC-derived pancreas in pancreas-deficient mice (Kobayashi et al., 2010). Since then, we and others have shown the generation of kidneys, thymus, and germ cells via blastocyst complementation using mouse and rat models (Kobayashi et al., 2021b, etc.). In our lab, we attempt to develop novel culture systems or embryo manipulation techniques applicable to blastocyst complementation.
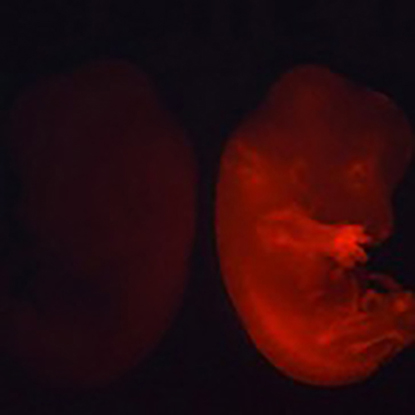

References
Kobayashi T et al., Generation of rat pancreas in mouse by interspecific blastocyst injection of pluripotent stem cells. Cell. 2010 Sep 3;142(5):787-99. doi: 10.1016/j.cell.2010.07.039.
Kobayashi T et al., Blastocyst complementation using Prdm14-deficient rats enables efficient germline transmission and generation of functional mouse spermatids in rats. Nat Commun. 2021b Feb 26;12(1):1328. doi: 10.1038/s41467-021-21557-x.




